|
Newly developed PET traces are more specific and allow for imaging biological processes, such as angiogenesis, hypoxia, proliferation, apoptosis and many others. F18-FDG limitations such as low specific targeting in PET imaging were an incentive for development of new PET tracers with special targeting capabilities. It is worth mentioning that first applications of FDG were focused on neurology and cardiac imaging, but its importance in oncology was realized on a later stage. In March 2000, the fluorine-18-labelled glucose or F-18 fluoro-2-deoxyglucose (F-18 FDG) received Food and Drug Administration FDA approval for usage to evaluate and diagnose oncology patients although the first production was in 1978 for neurological applications. 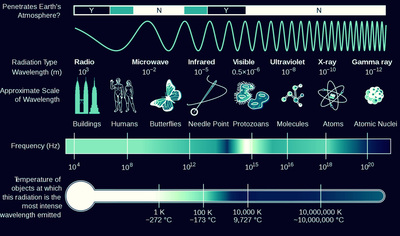
Radionuclides such as 201Tl, 67Ga, 111In-, 123I and 131I belong also to the same class and have several diagnostic or therapeutic applications. The former class of compounds are commonly labelled with 99mTc solution eluted from molybdenum-99 generators. Radiopharmaceuticals can be broadly classified into two different categories, single photon emitters that are commonly used in conventional gamma camera imaging examinations and positron emitter-based radiopharmaceuticals. Diagnostic radiopharmaceutical is a radioactive drug that is administered in a tracer quantity with no pharmacological effect on human body. The other good facet of nuclear medicine is its ability to target but treat critical diseases using therapeutic radionuclides. The diagnostic role is to interrogate valuable functional information about disease biochemistry not only on the cellular or subcellular level but also extend to extract molecular and genetic information. Nuclear medicine is a unique medical specialty that uses radiopharmaceuticals for diagnostic or therapeutic purposes. From the frequency distribution of stable isobars, isotopes and isotones, it has been concluded that even numbers of identical nucleons are more stable than odd numbers of the same nucleons (oddness of both Z and N tends to lower the nuclear binding energy). 1.6), the excess-neutron number is seen as the vertical distance between the stable nuclides and the diagonal N = Z line. Radioactive nuclei attempt to reach the stability by different modes of radioactive decay which is going to be discussed later. Radioactive nuclides surround the line of stability nuclides lying above the line are said to be ‘proton deficient’, whereas nuclides lying below the line are ‘neutron deficient’. From Z > 83 all heavier nuclides are unstable. Thus, the line of stability shows N ≈ 1.5 Z, that is heavy stable nuclides have nearly 50 % more neutrons than protons. For heavy nuclei, the coulomb repulsion between the protons is substantial, and extra neutrons are needed to supply additional binding energy to hold the nucleons together. 1.6 and closer look into light nuclei, the line of stability shows that neutron and proton numbers are equal ( N ≈ Z). 1.3.Ī major measure of nuclear stability is the neutron-proton ratio, as a result of the coulomb repulsion and exchange forces between them. 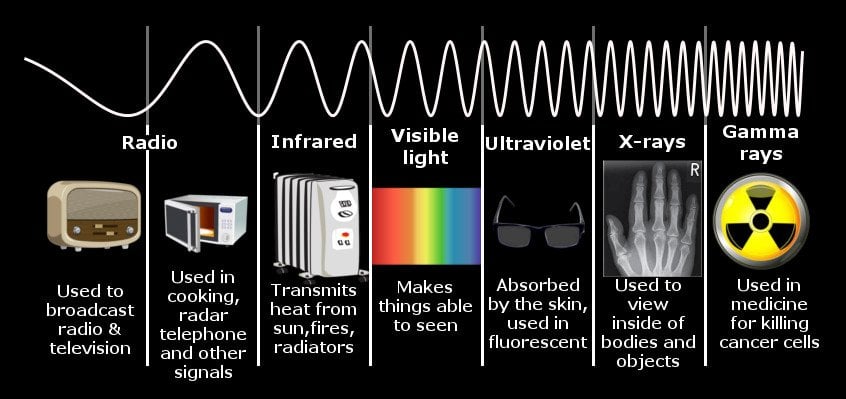
While this proposal was in 1927, the announcement of the understanding of the wave and particle aspects of matter was carried out in 1928. Then, Louis de Broglie proposed that electrons and other particles can behave as waves and has wavelength and frequencies. He stated that the emission of photoelectrons from metal plates does occur if and only if the incident photons on photoemissive plate have a threshold wavelength of energy that is able to liberate electrons from the metal. Another contribution came from the phenomenon of photoelectric effect proposed also by Einstein. Max Plank has stated that energy is transferred in a form of packets or quanta, while Albert Einstein thought of light as particle-like or localized in packets of discrete energy in contrast to its original definition as wave-like properties. It was not originated from a single scientist or revealed from only one experiment. 
The duality principle describes the elementary particles and electromagnetic radiation in terms of wave and particle-like characteristics.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
